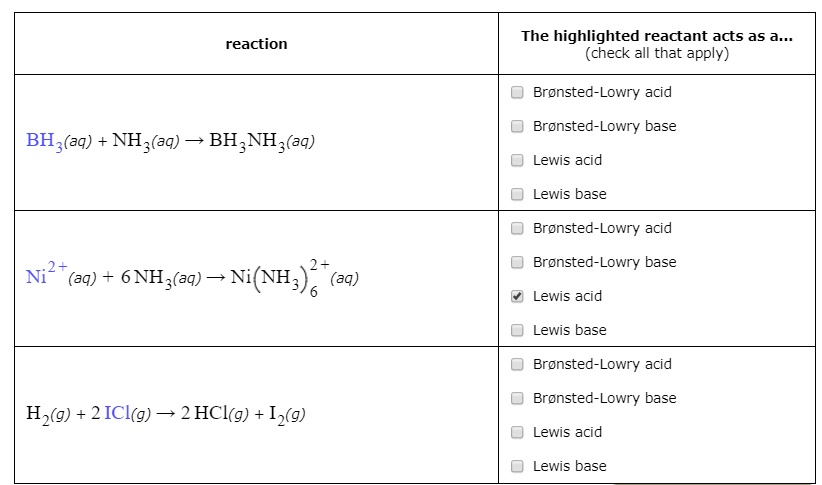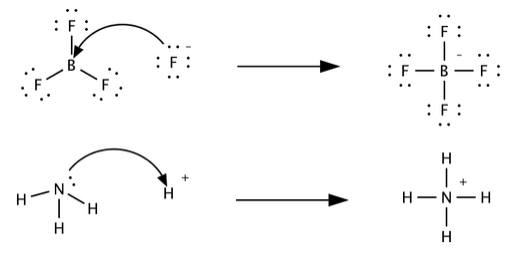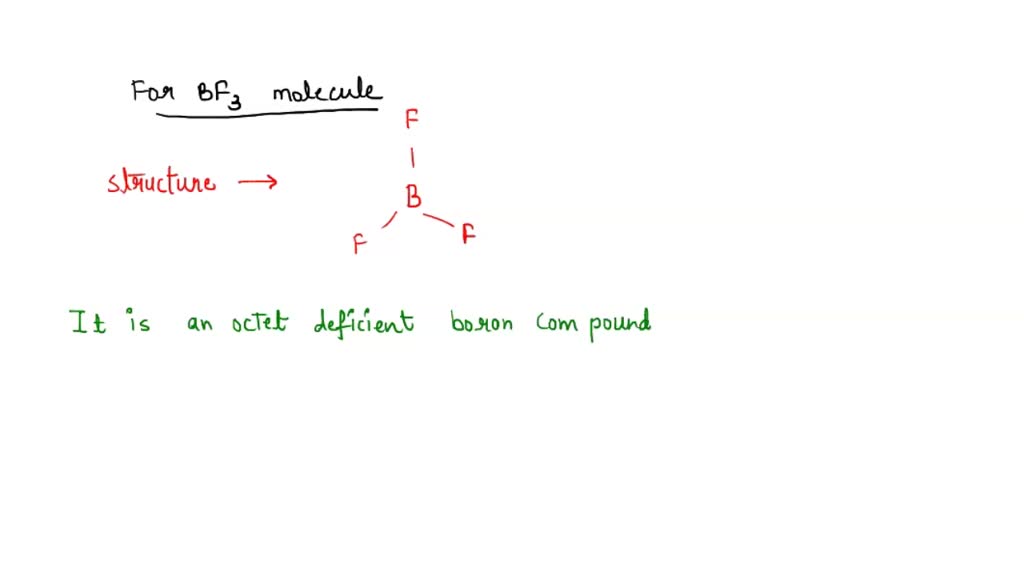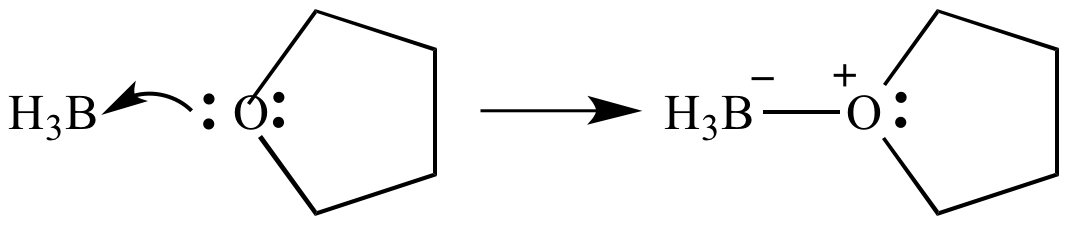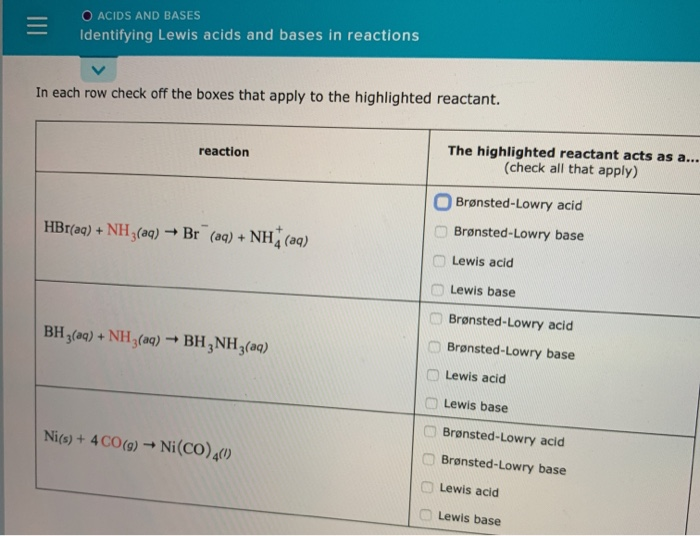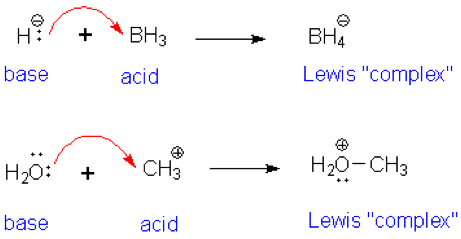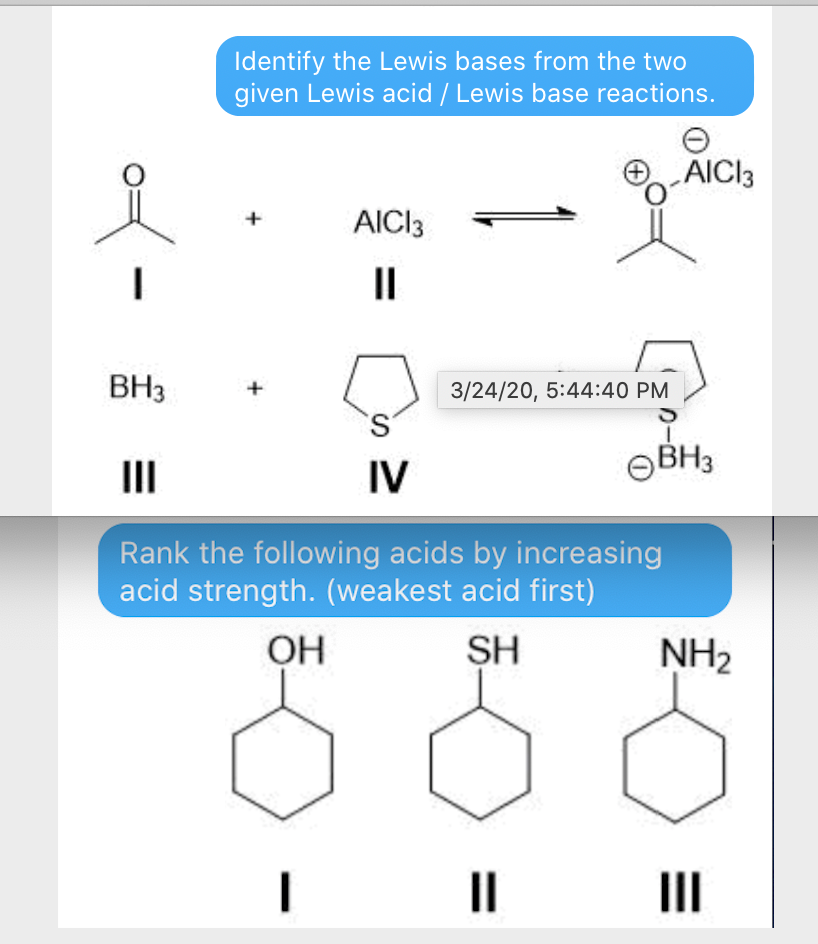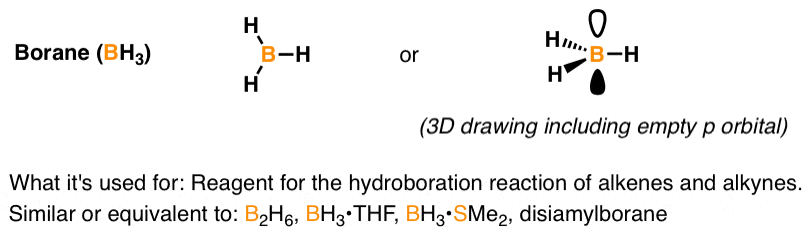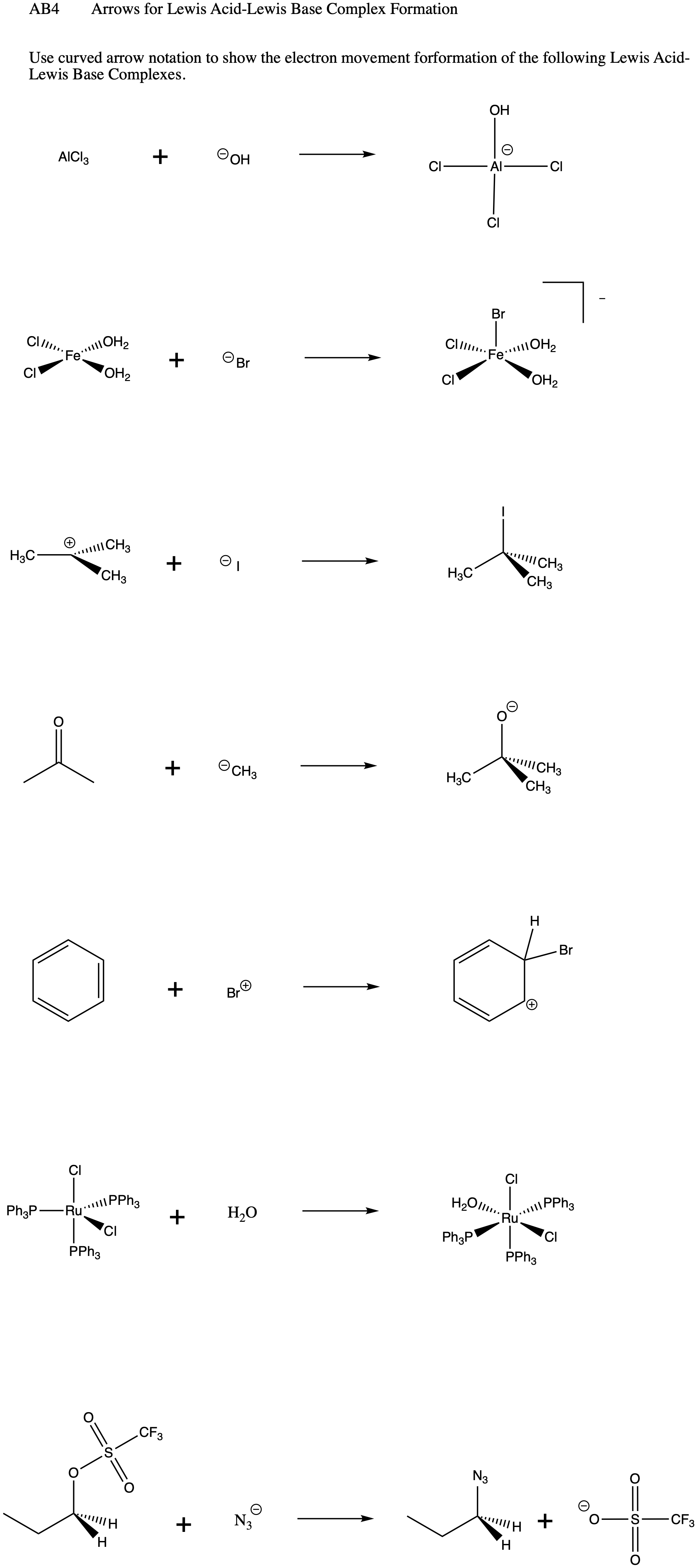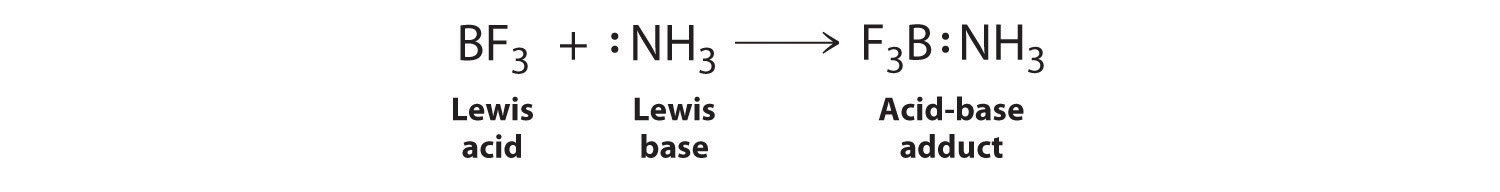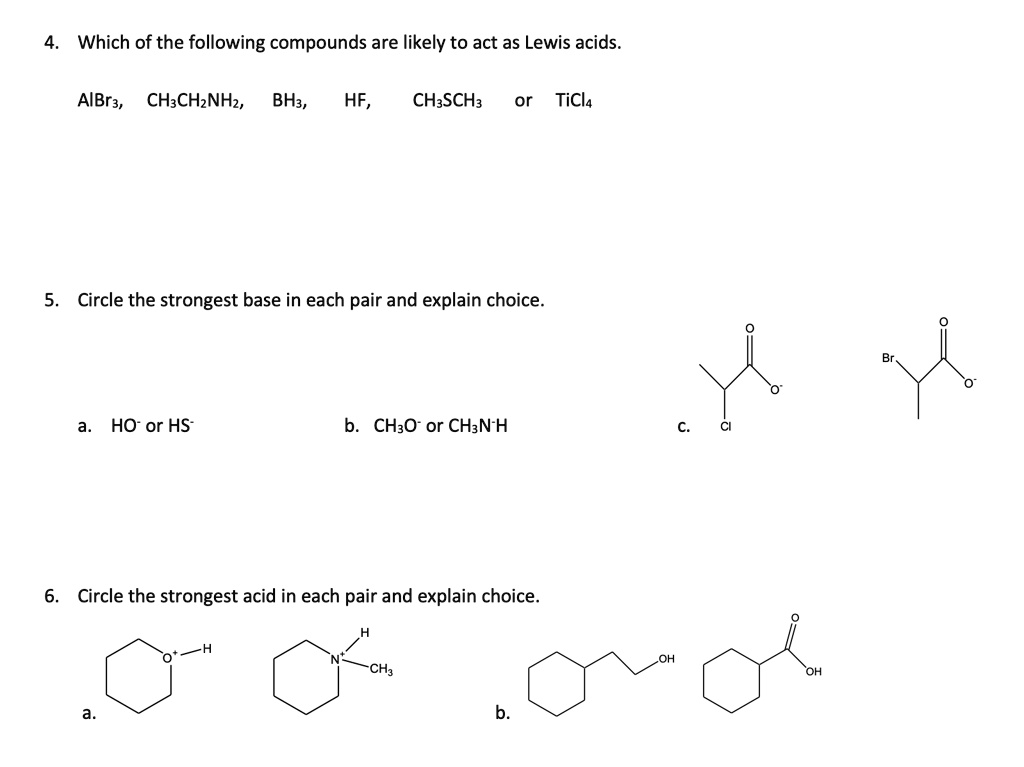
SOLVED: Which of the following compounds are likely to act as Lewis acids. AIBr3, CH3CHzNHz, BH3, HF, CH;SCH3 TiCla Circle the strongest base in each pair and explain choice HO or HS

Nature and Strength of Lewis Acid/Base Interaction in Boron and Nitrogen Trihalides - Rodrigues Silva - 2020 - Chemistry – An Asian Journal - Wiley Online Library